Science Seen Physicist and Time One author Colin Gillespie helps you understand your world.
Do Stars Make Buckyballs?
No doubt you never thought about this question. But suddenly it is hot science news. The answer that we get is: Yes. And it has interesting implications.
The story starts with the odd properties of carbon. (It’s our story too because they are the basis for life, not to mention much of our industry.) Carbon atoms love to make four strong bonds, especially with tetrahedral symmetry like pyramids. Pure carbon makes some solids, like soft graphite and hard diamond, and with other atoms such as hydrogen and oxygen it sponsors millions of compounds (solids, liquids, gases) with their own collective name: organic. They include long-chain molecules like DNA and short-chain ones like fats. You get the message: we see carbon compounds all around. But might other kinds of carbon compounds lurk in space? This question gets another Yes.
This question started with American astronomer Mary Lea Shane who, as a student in 1924, studied strange absorption lines in light from distant stars and claimed they showed there are organic chemicals in space. The answer 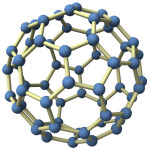 had to wait until this month. Using a new method to trap pure ions, a group at Basel University led by Prof. John Maier showed that buckminsterfullerene ions match two of the strange absorption lines. A hollow spherical shell of sixty carbon atoms, fullerene (aka Buckyballs) is named after American architect Richard Buckminster Fuller, promoter of the geodesic dome. Fullerene was discovered by Rice University chemists in 1985 and three of them got the 1996 Nobel Prize for it.
had to wait until this month. Using a new method to trap pure ions, a group at Basel University led by Prof. John Maier showed that buckminsterfullerene ions match two of the strange absorption lines. A hollow spherical shell of sixty carbon atoms, fullerene (aka Buckyballs) is named after American architect Richard Buckminster Fuller, promoter of the geodesic dome. Fullerene was discovered by Rice University chemists in 1985 and three of them got the 1996 Nobel Prize for it.
Thus a Swiss laboratory is the scene for solving the absorption-line problem but Mary Lea Shane’s question starts in space. Let’s go back and ask: So why would we find Buckyballs in space? Where do they come from? The answer seems almost certain: Stars make Buckyballs when they collapse. How can we be sure of this? Here is the reasoning.
For millions of years after the beginning there was no carbon in the universe, only hydrogen and helium and a trace of lithium. Though this was long ago, theory predicts it and measurements confirm it. Stars make all the carbon—in a process called nucleosynthesis—and eject it as carbon vapor into space late in their lives as they collapse under their own gravity. Again, well-established physics says this is the source of all the carbon atoms that showed up here on Earth. And we know that Buckyballs will grow from carbon vapor. Simply put, we know how stars can do the trick and we know of no other way. Next step might be to measure isotopic ratios to nail it down.
 Aside from the satisfaction that comes from finding the explanation what’s the message here? Well, let’s take another look at that C60 structure. What if IKEA sold it? (They don’t; though they do sell geodesic chairs.) You would unpack 150 pieces and click on B for Buckyball to find assembly instructions. Can you imagine putting it together? Each carbon atom’s fighting you to make four bonds, not three. And yet the real thing self-assembles out in space with no mistakes, by the gazillions. Herein (I say) lies a message. All the atoms we are made of come from the same source—collapsing stars—and get here the same way—through space. That leaves lots of time for self-assembling simpler molecules such as the building blocks of DNA and protein. We tend to think of life as being here on Earth. The message from those Buckyballs in space is this: We are astrobiology.
Aside from the satisfaction that comes from finding the explanation what’s the message here? Well, let’s take another look at that C60 structure. What if IKEA sold it? (They don’t; though they do sell geodesic chairs.) You would unpack 150 pieces and click on B for Buckyball to find assembly instructions. Can you imagine putting it together? Each carbon atom’s fighting you to make four bonds, not three. And yet the real thing self-assembles out in space with no mistakes, by the gazillions. Herein (I say) lies a message. All the atoms we are made of come from the same source—collapsing stars—and get here the same way—through space. That leaves lots of time for self-assembling simpler molecules such as the building blocks of DNA and protein. We tend to think of life as being here on Earth. The message from those Buckyballs in space is this: We are astrobiology.
Sources:
Ewen Campbell et al. (2015), “Laboratory confirmation of C60+ as carrier of two diffuse interstellar bands”, Nature, vol. 523, p. 322; http://www.nature.com/nature/journal/v523/n7560/full/nature14566.html
Other Materials:
Pascale Ehrenfreund & Bernard Foing (2015), “Fullerene solves an interstellar puzzle”, Nature, vol. 523, p. 296, 16 July; http://www.nature.com/nature/journal/v523/n7560/full/523296a.html
Image credits:
James Hedberg, CC BY-NC-SA 3.0
dpocius, http://www.instructables.com/id/Lighted-MALM-bed-an-all-IKEA-hack/step2/Bonus-hack-business-card/

No comments yet.